First Croatian Medicinal Mushroom Research:
The Testing of Dr Myko San Preparations
PRELIMINARY TESTING OF ANTI-CANCER PREPARATIONS OF THE COMPANY ‘DR MYKO SAN – HEALTH FROM MUSHROOMS’
Preliminary testing both in vitro and in vivo on the anti-cancer medicinal mushroom preparations LENTIFOM and LENTRAM, and their basic components, and toxicity tests of these preparations were carried out at the Department of Molecular Medicine, Rudjer Boskovic Institute in Zagreb. These preparations were produced by the company DR MYKO SAN – HEALTH FROM MUSHROOMS. Testing was conducted in 1999 and 2000 by Professor Mislav Jurin, PhD and Sinisa Ivankovic, MSc, with the technical support of Mrs. Nevenka Hirsl. Dr. Ivan Jakopovich participated in the study on behalf of DR MYKO SAN – HEALTH FROM MUSHROOMS.
EFFECT OF LENTIFOM AND LENTRAM ON FIBROSARCOMA CELL CULTURES
In January and February 2001, the team conducted new in vitro experiments, aimed at determining the effect of the concentrated forms of the LENTIFOM and LENTRAM preparations on fibrosarcoma (FsaR) and squamous cell carcinoma (SCC VII) cultures. It was proven that both preparations are very cytotoxic, such that the initial concentration, 50% in relation to the total quantity of nutritive media, applied for the cultivation of cancer cells (5 mL) and then in concentrations of 10% led to a complete reduction of the number of live cancer cells in the culture. Despite certain technical difficulties, we succeeded in photographing the results of the fibrosarcoma experiments:
Photo: Velizar Vesovic

Photo: Dr. Mirko Hadzija

Photo: Dr. Mirko Hadzija

Photo: Dr. Mirko Hadzija

Photo: Dr. Mirko Hadzija
![]()
IN VIVO EXPERIMENTS
Testing was conducted to see how long mice injected with tumor cells could survive after being given the extracts of some of the basic components of preparations of the mushrooms Lentinus edodes, Grifola frondosa and Coriolus versicolor, produced by the company Dr Myko San, in the concentrations in which they are found in the preparations. Mice (strain C3H/HZgr) were injected with approx. 5 x 105 cells of the very aggressive fibrosarcoma (FSar) tumor, or the squamous cell carcinoma (SCC VII). When the implanted tumors grew to an average size of 5 mm, the animals received a daily dose of 1 mL of mushrooms extracts in the abdominal cavity, over 15 days. Following this, the survival rate of each experimental group was examined, in comparison with the control group that did not receive any medicinal mushrooms extracts.
The most important results were:
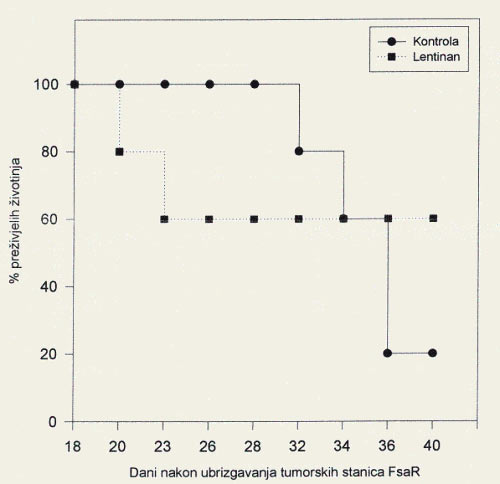
- On the final day of monitoring, 20% of the mice with fibrosarcoma in the control group were still alive, compared to 60% of the mice receiving Lentinus edodes mushroom extract (Figure 1).
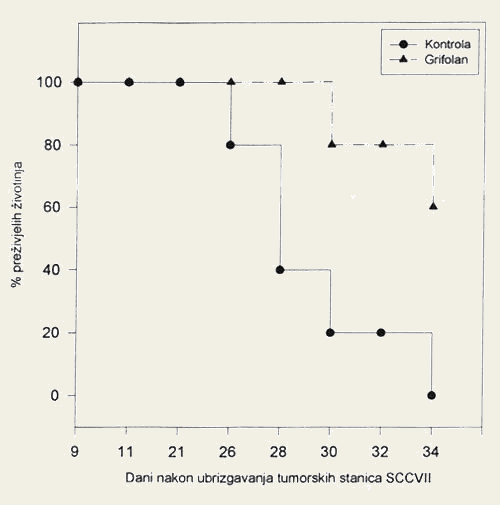
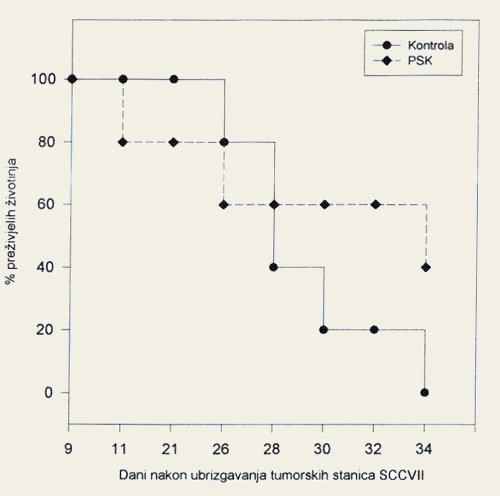
- On the final day of monitoring, none of the mice with squamous cell carcinoma in the control group were still alive, as compared to 60% of the mice receiving the extract of the mushroom Grifola frondosa (Figure 2).
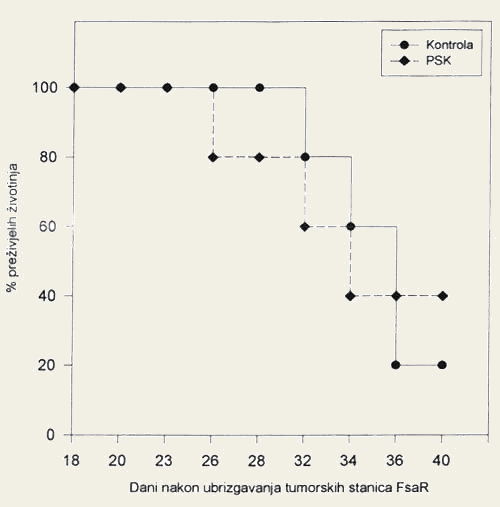
- On the final day of monitoring, 20% of the mice with fibrosarcoma in the control group were still alive, as compared to 40% of the mice receiving the extract of the mushroom Coriolus versicolor (Figure 3).
- On the final day of monitoring, none of the mice with squamous cell carcinoma in the control group were still alive, as compared to 40% of the mice receiving the extract of the mushroom Coriolus versicolor (Figure 4).
![]()
IN VITRO EXPERIMENTS
Testing of the anti-tumor preparations and some of their basic components was carried out on cell cultures in vitro in late January and early February 2000.
The first experiment examined the activity of various concentrations of extracts of the mushrooms Coriolus versicolor, Grifola frondosa and Lentinus edodes, produced by the company Dr Myko San – Health from Mushrooms, on cell cultures of two very malignant tumors – squamous cell carcinoma (SCC VII) and fibrosarcoma (FSar), in comparison to control cell cultures. Cells of squamous cell carcinoma and fibrosarcoma, were taken from cell cultures in a concentration of 2 x 105 live tumor cells/mL, and were cultivated in an incubator for 72 hours, with the addition of 100 µL of the extracts of the above mushrooms (no extracts were added to the control). During the final 24 hours of incubation, radioactive thymidine was added to the cultures. Following completion of cultivation, the intensity of radioactivity of the incorporated thymidine was measured. The smaller the counts per minute (CPM), the high the share of destroyed tumor cells in the culture. The most significant results were as follows:
- In comparison to a CPM of approx. 13,800-16,500 in control cultures, the highest concentrations of extracts of the mushroom Coriolus versicolor reduced the CPM in squamous cell carcinoma cultures to 522, and to only 188 in fibrosarcoma cultures.
- In comparison to the above CPM in normal cultures, the highest concentrations of extracts of the mushroomGrifola frondosa reduced the CPM in squamous cell carcinoma cultures to 274, while the median concentration of this extract reduced the CPM in fibrosarcoma cultures to 423.
- Especially interesting results were seen with the extracts of the mushroom Lentinus edodes. The weakest concentrations reduced the CPM in squamous cell carcinoma to 207 in comparison to the control, while the highest concentration of the extract reduced the CPM in fibrosarcoma cell cultures to only 170.
| 21.1.2000. | SCC VII | FSar |
|---|---|---|
| CV 25 | 862 | 9 359 |
| CV 50 | 834 | 1 404 |
| CV 100 | 522 | 188 |
| GF 25 | 1 143 | 4 793 |
| GF 50 | 1 102 | 423 |
| GF 100 | 274 | 435 |
| LE 25 | 207 | 224 |
| LE 50 | 273 | 200 |
| LE 100 | 271 | 170 |
| CONTROL | 16 459 | 14 160 |
| CONTROL | 13 792 | 14 139 |
| CONTROL | 14 376 | 14 989 |
The second experiment examined the activity of different concentrations of standard and concentrated extracts of the mushroom Coriolus versicolor, produced by the company Dr Myko San, and different concentrations of the preparations LENTIFOM and LENTRAM, standard products of the same company, in their standard and concentrated forms on cell cultures of three very malignant tumors: squamous cell carcinoma (SCC VII), fibrosarcoma (FsaR) and melanoma (B16F10), in comparison to control cell cultures. The most significant results were as follows:
- In this experiment, a particularly resilient clone of squamous cell carcinoma appeared, and the extracts of the mushrooms Coriolus versicolor and the products LENTIFOM and LENTRAM could not inhibit its growth. However, in comparison to a CPM of approx. 2070-2890 in the control cultures of this carcinoma, the concentrated form of LENTIFOM, in the strongest of the three applied concentrations, reduced the CPM in the cell culture of this carcinoma to 407.
- Control cell cultures of fibrosarcoma had a CPM of approx. 53,900-61,600. The standard form of LENTIFOM, in the strongest of the three applied concentrations, reduced the CPM in the fibrosarcoma cell culture to only 90, while the concentrated form of LENTIFOM, in the highest concentration reduced it to 233, and in the medium concentration to 131. LENTRAM gave similar results: the standard form in the strongest concentration reduced the CPM to 168, while the concentrated form of LENTRAM in reduced the CPM in the fibrosarcoma cell culture to 127 with the highest concentration, and to 171 with the middle concentration.
- In the melanoma cell cultures, the control cultures had a CPM of approx. 50,140 to 52,100. The standard form of LENTIFOM, in the strongest of the three applied concentrations, reduced the CPM in melanoma cell cultures to 1808, while the concentrated form of LENTIFOM, in the middle concentration, reduced the CPM to 3590. LENTRAM was less efficient in most concentrations of the standard and concentrated form, though the highest concentration of the concentrated form reduced the CPM in the melanoma cell culture to only 263.
- As a further control in this experiment, the preparations were also applied to normal cells (fibroblasts), which had a CPM of approx. 1460-2160 in the control samples. With the exception of the concentrated form of LENTIFOM in the highest concentration (CPM of only 260), and standard LENTIFOM in the highest concentration (CPM of 1612), the standard and concentrated extracts of the mushrooms Coriolus versicolor, and the preparations of LENTIFOM and LENTRAM in the standard and concentrated form, either moderately or strongly stimulated the growth of normal fibroblasts in all the applied concentrations.
| 9.2.2000. | V79 | SCC VII | FSar | B16 F10 |
|---|---|---|---|---|
| CV 25 | 7 069 | 3 851 | 14 297 | 38 235 |
| CV 12,5 | 3 41 | 3 027 | 52 338 | 48 307 |
| CV 6,25 | 2 772 | 2 686 | 60 085 | 49 096 |
| CV1 25 | 10 208 | 7 435 | 6 808 | 22 470 |
| CV1 12,5 | 7 884 | 5 418 | 20 265 | 34 463 |
| CV1 6,25 | 3 580 | 3 326 | 53 888 | 41 778 |
| LF 25 | 1 612 | 3 222 | 90 | 1 808 |
| LF 12,5 | 4 781 | 7 189 | 464 | 17 332 |
| LF 6,25 | 5 466 | 7 478 | 5 185 | 35 479 |
| LF1 25 | 260 | 467 | 233 | 25 119 |
| LF1 12,5 | 2 340 | 3 217 | 131 | 3 590 |
| LF1 6,25 | 5 580 | 6 377 | 1 596 | 17 391 |
| LENT 25 | 11 777 | 17 740 | 168 | 13 525 |
| LENT 12,5 | 11 685 | 15 678 | 7 764 | 37 068 |
| LENT 6,25 | 6 420 | 7 451 | 28 823 | 40 188 |
| LENT1 25 | 12 356 | 6 403 | 127 | 263 |
| LENT1 12,5 | 8 879 | 16 923 | 171 | 24 352 |
| LENT1 6,25 | 7 715 | 12 419 | 17 516 | 43 360 |
| CONTROL | 2 158 | 2 889 | 61 619 | 52 078 |
| CONTROL | 1 460 | 2 067 | 59 723 | 51 313 |
| CONTROL | 1975 | 2 061 | 53 904 | 50 142 |
TOXICITY TESTS
Prior to testing the effects of LENTIFOM and LENTRAM at the Department of Molecular Medicine of the Rudjer Boskovic Institute in Zagreb, in 1999 they were tested for possible toxic effects. The effect of each preparation was tested into 3 groups of 10 mice each. For a duration of 41 days, the mice received LENTIFOM or LENTRAM, with one group receiving the preparation in undiluted form, another receiving the preparation diluted with water in the ratio 1:5, and the third received the preparation diluted with water in the ratio 1:25. For a duration of 11 days, the mice received a dose of 0.5 mL of the preparation in the abdominal cavity (intraperitoneally). Throughout the experiment, body weight, possible side effects and survival rates of the mice were recorded.
The results were as follows: Body weight of mice either grew or declined to some extent, or remained the same. All animals survived the 41 day experiment, and no side effects were observed at any time. Thus, it was confirmed that neither LENTIFOM nor LENTRAM, either diluted or undiluted, had any toxic effects.
Also read more about Myko San and Rudjer Boskovic Institute research project and published work, The Influence of Medicinal Mushroom Preparation on Mouse Tumors.
![]()
